|
To convert 25☌ to a temperature in Kelvin (K): If you have a temperature in ☌ we can just add 273.15 to arrive at a temperature in Kelvin (K): This makes converting between temperatures in ☌ and K straight-forward. The divisions in the Celsius scale and the Kelvin scale are the same size:įor example, the difference between the temperature at which water boils and freezes at 1 atm:Ī difference of 1☌ is the same as a difference of 1K, the only difference is the starting point (at 1 atm, the freezing point of water is 0☌ or 273.15 K). Let's compare three common temperature scales: Kelvin, Celsius and Fahrenheit: No ads = no money for us = no free stuff for you! Temperature Conversions: Kelvin, Celsius, Fahrenheit To convert a temperature in Celsius (☌) to a temperature in Fahrenheit (☏).To convert a temperature in Fahrenheit (☏) to a temperature in Celsius (☌).To convert a temperature in Kelvin (K) to a temperature in Celsius (☌).To convert a temperature in Celsius (☌) to a temperature in Kelvin (K). 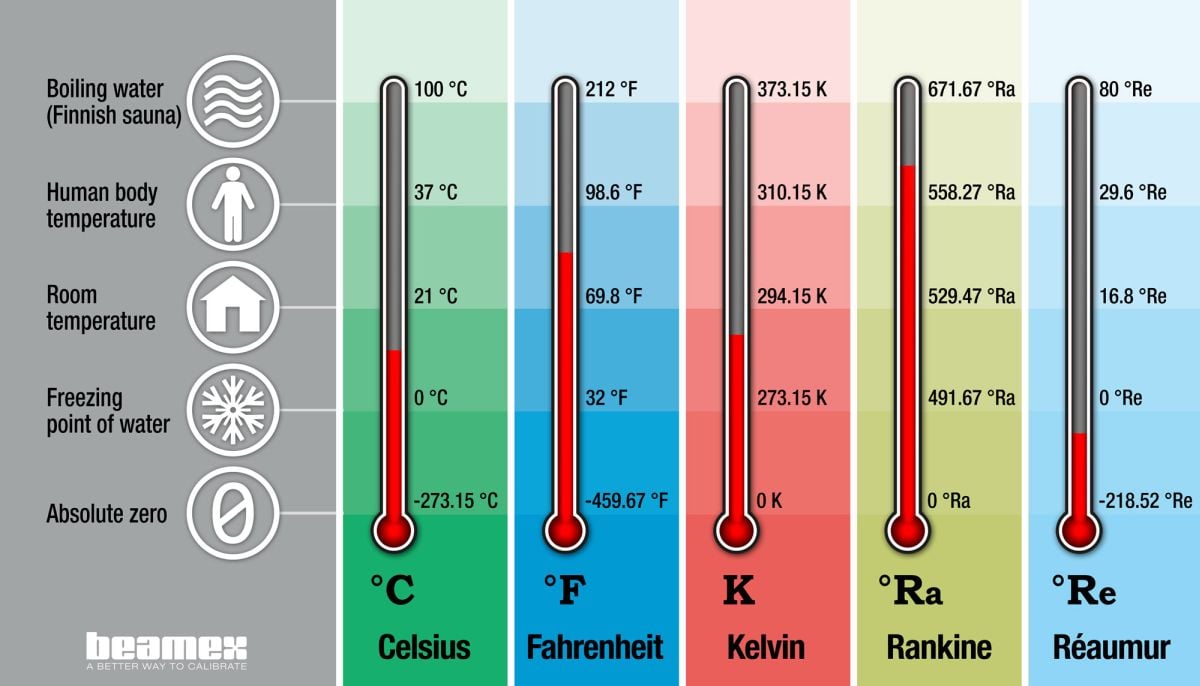
⚛ boiling point of water as 100.00☌ at 1 atm pressureĪ dfference in temperature of 1☌ = a difference of 1KĪ dfference in temperature of 1☏ = a difference of 5/ 9☌ = a difference of 5/ 9K ⚛ freezing point of water as 0.00☌ at 1 atm pressure ⚛ a triple-point temperature of water of 0.01☌ at 1 atm pressure ⚛ The temperature at which solid, liquid and gaseous water can coexist indefinitely (the triple-point temperature of water) is assigned a value of 273.16 K ⚛ The zero point is absolute zero, the lowest temperature theoretically obtainable. Kelvin Scale is based on the following fixed points:.⚛ Fahrenheit Scale based on degrees Fahrenheit (☏), commonly used in North America. ⚛ Celsius Scale based on degrees Celsius (☌), closely related to the Centigrade scale. ⚛ Kelvin Scale based on the kelvin unit (K) is the SI unit of temperature. Several temperature scales are in common use:. 
When an object loses energy, kinetic energy of the particles decreases and is observed as a decrease in temperature.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |


 RSS Feed
RSS Feed
